Current issue
Accepted manuscript
About the Journal
Scientific Council
Editorial Board
Regulatory and archival policy
Code of publishing ethics
Publisher
Information about the processing of personal data in relation to cookies and newsletter subscription
Archive
For Authors
For Reviewers
Contact
Reviewers
Annals reviewers in 2025
Annals reviewers in 2024
Annals reviewers in 2023
Annals reviewers in 2022
Annals reviewers in 2021
Annals reviewers in 2020
Annals reviewers in 2019
Annals reviewers in 2018
Annals reviewers in 2017
Annals reviewers in 2016
Annals reviewers in 2015
Annals reviewers in 2014
Annals reviewers in 2013
Annals reviewers in 2012
Links
Sklep Wydawnictwa SUM
Biblioteka Główna SUM
Śląski Uniwersytet Medyczny w Katowicach
Privacy policy
Accessibility statement
Reviewers
Annals reviewers in 2025
Annals reviewers in 2024
Annals reviewers in 2023
Annals reviewers in 2022
Annals reviewers in 2021
Annals reviewers in 2020
Annals reviewers in 2019
Annals reviewers in 2018
Annals reviewers in 2017
Annals reviewers in 2016
Annals reviewers in 2015
Annals reviewers in 2014
Annals reviewers in 2013
Annals reviewers in 2012
The analysis of sodium butyrate content in various dietary supplements available on the Polish market
1
Department of Internal Medicine and Clinical Pharmacology, Faculty of Medical Sciences in Katowice, Medical University of Silesia, Katowice, Poland
Corresponding author
Maciej Borówka
Klinika Chorób Wewnętrznych i Farmakologii Klinicznej, ul. Medyków 14, 40-752 Katowice, Uniwersyteckie Centrum Kliniczne im. prof. K. Gibińskiego ŚUM
Klinika Chorób Wewnętrznych i Farmakologii Klinicznej, ul. Medyków 14, 40-752 Katowice, Uniwersyteckie Centrum Kliniczne im. prof. K. Gibińskiego ŚUM
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Natrium butyrate is a chemical compound which seems to have several beneficial properties, which alleviate signs and symptoms of gastrointestinal diseases. As a result, it has become a frequent component of therapeutic regimens. However, all of the oral products containing natrium butyrate available on the Polish market are registered as dietary supplements and none of them is approved for use as a medication, which might raise concerns about their quality.
Material and methods:
Natrium butyrate from eight different dietary supplements available on the Polish market in April 2024 was extracted with diethyl ether. Subsequently, diethyl ether was evaporated and remnants were dissolved in methanol in order to perform high performance liquid chromatography (HPLC). Obtained solutions were passed through a chromatography column to assess exact content of natrium butyrate in each sample. Finally, obtained peaks were compared with standard samples and the content of natrium butyrate in each sample was calculated.
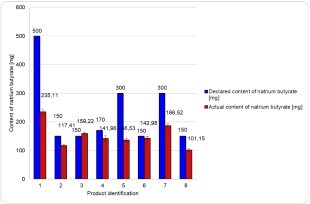
Results:
Sodium butyrate content was detected in each of the analyzed samples. However, the percentage ratio of the amount detected to that declared by the producers varied from 45.51% to 106.15%.
Conclusions:
In the majority of cases, analyzed dietary supplements contained natrium butyrate in quantities similar to those declared by manufacturers; however, compliance with the manufacturer's declaration varied between specific products, highlighting the need for further research in this area.
Natrium butyrate is a chemical compound which seems to have several beneficial properties, which alleviate signs and symptoms of gastrointestinal diseases. As a result, it has become a frequent component of therapeutic regimens. However, all of the oral products containing natrium butyrate available on the Polish market are registered as dietary supplements and none of them is approved for use as a medication, which might raise concerns about their quality.
Material and methods:
Natrium butyrate from eight different dietary supplements available on the Polish market in April 2024 was extracted with diethyl ether. Subsequently, diethyl ether was evaporated and remnants were dissolved in methanol in order to perform high performance liquid chromatography (HPLC). Obtained solutions were passed through a chromatography column to assess exact content of natrium butyrate in each sample. Finally, obtained peaks were compared with standard samples and the content of natrium butyrate in each sample was calculated.
Results:
Sodium butyrate content was detected in each of the analyzed samples. However, the percentage ratio of the amount detected to that declared by the producers varied from 45.51% to 106.15%.
Conclusions:
In the majority of cases, analyzed dietary supplements contained natrium butyrate in quantities similar to those declared by manufacturers; however, compliance with the manufacturer's declaration varied between specific products, highlighting the need for further research in this area.
FUNDING
This research received no external funding. The authors have no relevant financial or non-financial interests to disclose.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
REFERENCES (17)
1.
Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016;7(3):189–200. doi: 10.1080/19490976.2015.1134082.
2.
Wong JMW, de Souza R, Kendall CWC, Emam A, Jenkins DJA. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40(3):235–243. doi: 10.1097/00004836-200603000-00015.
3.
Lewandowski K, Kaniewska M, Karłowicz K, Rosołowski M, Rydzewska G. The effectiveness of microencapsulated sodium butyrate at reducing symptoms in patients with irritable bowel syndrome. Prz Gastroenterol. 2022;17(1):28–34. doi: 10.5114/pg.2021.112681.
4.
Scarpellini E, Lauritano EC, Lupascu A, Petruzzellis C, Novi ML, Roccarina D, et al. Efficacy of butyrate in the treatment of diarrhoea-predominant irritable bowel syndrome. Dig Liver Dis Suppl. 2007;1(1):19–22. doi: 10.1016/S1594-5804(08)60006-6.
5.
Facchin S, Vitulo N, Calgaro M, Buda A, Romualdi C, Pohl D, et al. Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel disease. Neurogastroenterol Motil. 2020;32(10):e13914. doi: 10.1111/nmo.13914.
6.
Recharla N, Geesala R, Shi XZ. Gut microbial metabolite butyrate and its therapeutic role in inflammatory bowel disease: A literature review. Nutrients. 2023;15(10):2275. doi: 10.3390/nu15102275.
7.
Mayorga-Ramos A, Barba-Ostria C, Simancas-Racines D, Guamán LP. Protective role of butyrate in obesity and diabetes: New insights. Front Nutr. 2022;9:1067647. doi: 10.3389/fnut.2022.1067647.
8.
Peng K, Dong W, Luo T, Tang H, Zhu W, Huang Y, et al. Butyrate and obesity: Current research status and future prospect. Front Endocrinol (Lausanne). 2023;14:1098881. doi: 10.3389/fendo.2023.1098881.
9.
Obwieszczenie Ministra Zdrowia z dnia 23 grudnia 2022 r. w sprawie ogłoszenia jednolitego tekstu rozporządzenia Ministra Zdrowia w sprawie składu oraz oznakowania suplementów diety. Dz.U. 2023 poz. 79.
10.
Ustawa z dnia 6 września 2001 r. Prawo farmaceutyczne. Dz.U. 2001 nr 126 poz. 1381.
11.
Dwyer JT, Coates PM, Smith MJ. Dietary supplements: regulatory challenges and research resources. Nutrients. 2018;10(1):41. doi: 10.3390/nu10010041.
12.
Ronis MJJ, Pedersen KB, Watt J. Adverse effects of nutraceuticals and dietary supplements. Annu Rev Pharmacol Toxicol. 2018;58:583–601. doi: 10.1146/annurev-pharmtox-010617-052844.
13.
De Baere S, Eeckhaut V, Steppe M, De Maesschalck C, De Backer P, Van Immerseel F, et al. Development of a HPLC-UV method for the quantitative determination of four short-chain fatty acids and lactic acid produced by intestinal bacteria during in vitro fermentation. J Pharm Biomed Anal. 2013;80:107–115. doi: 10.1016/j.jpba.2013.02.032.
14.
Eberhart BL 2nd, Wilson AS, O’Keefe SJD, Ramaboli MC, Nesengani LT. A simplified method for the quantitation of short-chain fatty acids in human stool. Anal Biochem. 2021;612:114016. doi: 10.1016/j.ab.2020.114016.
15.
Maćkiewicz E, Klimaszewska M, Albińska J, Szynkowska-Jóźwik MI. Evaluation of the elemental composition of dietary supplements containing iron available on the Polish market using ICP-OES, FAAS and CVAAS techniques. Molecules. 2025;30(23):4511. doi: 10.3390/molecules30234511.
16.
Wołonkiewicz K, Podolak I, Paśko P, Galanty A. Analysis of caffeine content in weight-loss dietary supplements. Farm Pol. 2019;75(2):53–56. doi: 10.32383/farmpol/116302.
17.
Puścion-Jakubik A, Bartosiewicz N, Socha K. Is the Magnesium Content in Food Supplements Consistent with the Manufacturers’ Declarations? Nutrients. 2021;13(10):3416. doi: 10.3390/nu13103416.
The Medical University of Silesia in Katowice, as the Operator of the annales.sum.edu.pl website, processes personal data collected when visiting the website. The function of obtaining information about Users and their behavior is carried out by voluntarily entered information in forms, saving cookies in end devices, as well as by collecting web server logs, which are in the possession of the website Operator. Data, including cookies, are used to provide services in accordance with the Privacy policy.
You can consent to the processing of data for these purposes, refuse consent or access more detailed information.
You can consent to the processing of data for these purposes, refuse consent or access more detailed information.